- Home >
- Learning centre >
- Science blog >
- Challenge Testing: Do We Need to Count Colonies to Prove Stability?
Challenge Testing: Do We Need to Count Colonies to Prove Stability?
9th April 2026

What challenge testing is trying to answer
Challenge testing, also known as preservative efficacy testing (PET) and antimicrobial effectiveness testing (AET), sits at the heart of product safety. Whether in cosmetics, pharmaceuticals, or food, the question is always the same:
Will this formulation prevent microbial growth over time?
Across industries, standardized methods such as those described in the European Pharmacopoeia (EP 5.1.3) and ISO 11930 for cosmetics define how antimicrobial effectiveness should be evaluated. The process is well established. A known concentration of microorganisms is introduced into a product, and the formulation is monitored over time. At defined intervals, often over days or weeks and sometimes up to 28 days, samples are taken, diluted, plated, and counted to determine whether microbial populations decline, persist, or grow.
These methods have stood the test of time. They are trusted, accepted, and deeply embedded in regulatory frameworks. But they are also slow, labor-intensive, and fundamentally indirect.
Are colonies the right readout?
A typical challenge test requires repeated sampling, serial dilutions, agar plating, incubation, and manual colony counting. Every time point adds another layer of work. Every plate introduces variability. Every readout is delayed by the need for visible growth. When products are opaque, such as creams, lotions, emulsions, or complex food matrices, even basic measurements like turbidity become unreliable or impossible.
From an operational standpoint, it is a heavy process. From a scientific standpoint, it is a proxy.
What we are really asking in challenge testing is not how many colonies we can count after incubation. The real question is much simpler:
Can microorganisms grow in this product?
Everything else, plating, dilution, incubation, is a way of answering that question indirectly. And that raises an important possibility: what if we could answer it directly?
Microorganisms do not need to form visible colonies to be active. Long before a colony appears on an agar plate, cells are metabolizing, adapting, and responding to their environment. If a preservative system fails, the earliest signal is not a colony, it is an increase in metabolic activity.
This is where biocalorimetry offers a fundamentally different perspective.
microbial CHALLENGE TESTING WITH BIOCALORIMETRY
Instead of waiting for visible growth, biocalorimetry measures metabolic heat, a direct and continuous signal of microbial activity. If bacteria are alive and begin to proliferate, their metabolism increases and produces a measurable thermal signal. If a formulation successfully inhibits growth, that signal remains flat or declines. In the context of challenge testing, this reframes the entire workflow.
Rather than repeatedly sampling and plating over weeks, the product can be inoculated once, sealed in measurement vials, and monitored continuously. There is no need to rely on optical clarity. Opaque creams, lotions, and complex food products can be tested without modification. There is no need for serial dilutions or daily plate checks. The system simply observes whether metabolic activity increases over time.
If it does, the preservative system is failing.
If it does not, the formulation is doing its job.
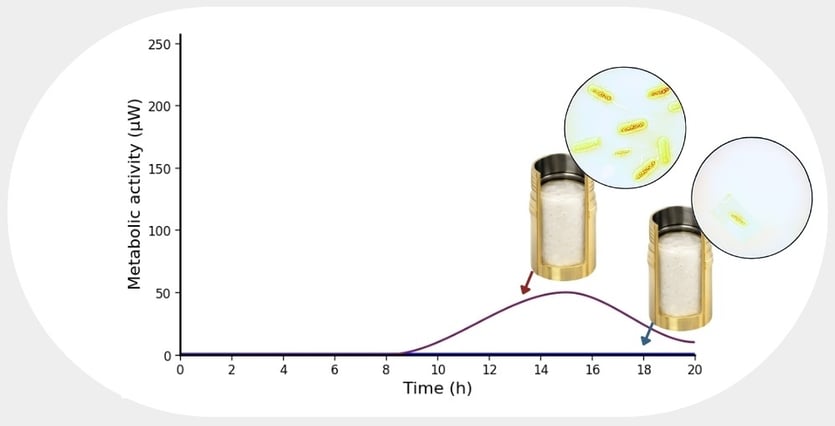
The simplicity of the question is finally matched by the simplicity of the readout. This approach also captures phenomena that traditional methods can miss. In some cases, microorganisms exposed to preservatives enter a viable but non-culturable state. They may not form colonies on agar plates, leading to an apparent reduction in CFU counts, yet they remain metabolically active and potentially capable of recovery under favorable conditions. Biocalorimetry detects this activity. It does not depend on culturability on plates. It measures life directly.
From a practical perspective, the implications are significant.
MORE EFFICIENT MICROBIAL TESTING FOR FASTER formulation and product development
Challenge testing today consumes time, labor, and consumables. It requires trained personnel to execute repetitive workflows and interpret results. It introduces delays between sampling and readout. And it often forces compromises when working with complex or opaque products.
By contrast, a metabolic monitoring approach simplifies the process to its essentials. Add the challenge organisms to the product. Place the sample in the instrument. Monitor continuously. Let the data reveal whether growth occurs.
This is not about inventing a new test. The principle of challenge testing remains unchanged: introduce microorganisms and observe whether they survive and proliferate. What changes is how we observe it:
-
A shift from indirect to direct measurement.
-
From endpoint snapshots to continuous insights.
-
From labor-intensive workflows to automated, high-resolution monitoring.
For laboratories and product developers, this opens up new possibilities. Faster iteration of formulations. Reduced hands-on time. Improved consistency. And the ability to test products as they are, without adapting them to fit the limitations of traditional assays.
Challenge testing will always be a cornerstone of product safety. But as products become more complex and development timelines accelerate, the methods used to evaluate them must evolve as well.
The question has always been simple: will microbes grow? It may be time to answer it in a more efficient way.
Learn more
Read more about microbial testing with the calScreener biocalorimeter system.

STAY UP-TO-DATE
Subscribe to our newsletter and stay up to date with the latest news and updates